
U.S. health authorities in Louisiana have confirmed that at least five people have died this year as a result of infections caused by the bacterium Vibrio vulnificus, commonly known as the “flesh-eating bacteria.” In total, 26 cases of infection have been reported in the region, most linked to open wounds
Read more: “Louisiana Reports Five Deaths Linked to Deadly Flesh-Eating Bacteria”

A new study published in the prestigious journal JAMA has revealed a significant deterioration in children’s health across the United States between 2007 and 2023, raising serious concerns in the medical and educational communities. The research, conducted by specialists from the Children’s Hospital of
Read more: “Study warns of worsening children’s health across the United States”
The Advisory Committee on Immunization Practices (ACIP) of the U.S. Centers for Disease Control and Prevention (CDC) has recommended that COVID-19 vaccination should no longer be a universal requirement for everyone over the age of six months, but rather an individualized decision. According to the
Read more: “CDC shifts vaccination strategy, leaving COVID-19 shots to patients”
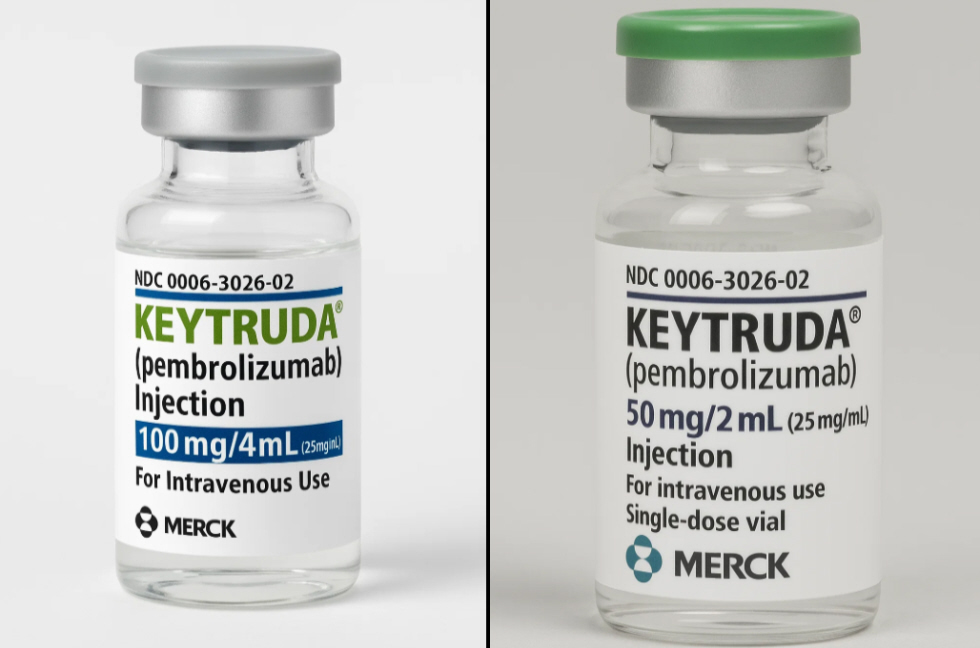
The U.S. Food and Drug Administration (FDA) has approved a new subcutaneous formulation of Keytruda, Merck’s flagship immunotherapy for cancer. This version, called Keytruda Qlex, represents a significant advancement in how the treatment is administered, as it allows patients to receive their dose
Read more: "FDA Approves Subcutaneous Version of Keytruda for Cancer Patients"


